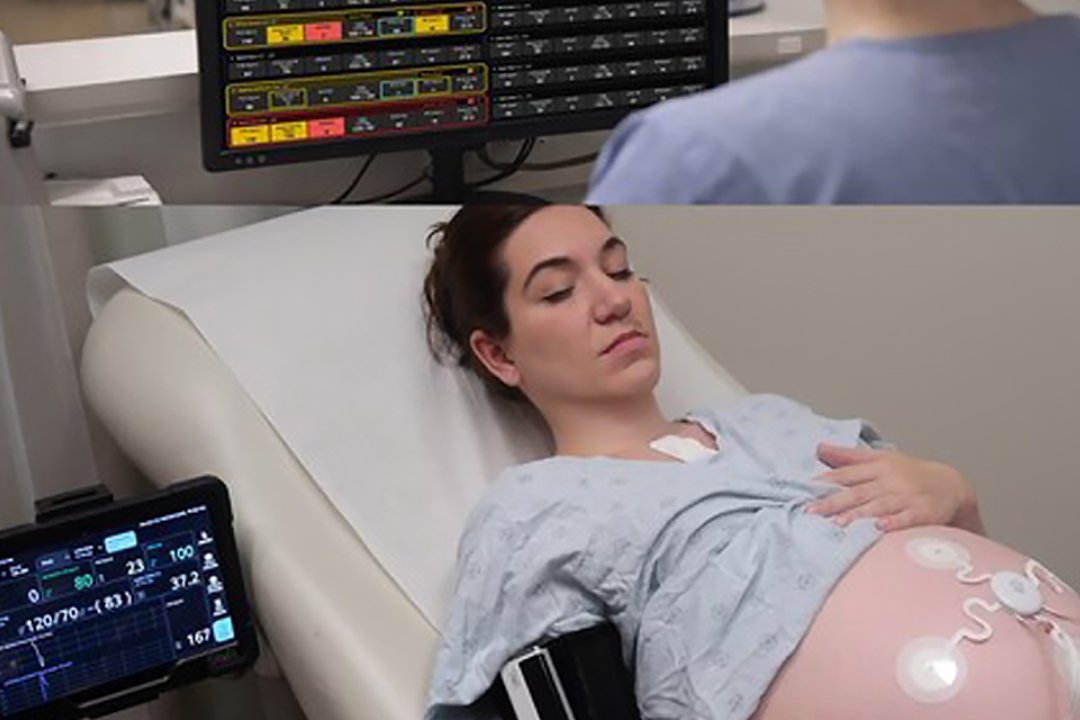
Sibel Health has announced that its ANNE Maternal platform has received 510(k) clearance from the US Food and Drug Administration. The Chicago based company developed the system as a fully wireless solution for monitoring both mothers and foetuses simultaneously. This technology represents a shift from traditional wired equipment that often keeps patients confined to hospital beds during labour.
The platform utilises flexible sensors that stick to the skin to track essential vitals. These include heart rate, respiratory rate, skin temperature and blood oxygen saturation. It also monitors foetal heart rates and uterine activity. The system includes a digital early warning feature called MEOWS to alert medical staff if a patient’s condition changes. This wireless approach is intended to provide comfort and mobility for patients while maintaining high clinical standards.
This regulatory approval comes at a time when maternal health figures in the US are under scrutiny. Current data shows 22 deaths occur per 100,000 live births in the United States, which is the highest rate among high-income countries. Sibel Health aims to make continuous monitoring a standard of care to help reduce these numbers.
The development of the platform was supported by a 17.5 million dollar grant from the Gates Foundation. Steve Xu, MD, CEO and Co-Founder of Sibel Health, said: “What makes this platform truly unique is its dual mandate: it meets the rigorous clinical standards demanded by the FDA while also being designed for deployment in the world’s most resource-limited settings. That convergence — high-income market validation and global health impact in a single platform — is what we’ve been building toward since day one. We are deeply grateful to the Gates Foundation, whose continued investment has made this product possible, and to our global partners whose clinical expertise and dedication to global health have been indispensable. Together, we are closer than ever to making continuous maternal monitoring a universal standard of care.”
The ANNE Maternal system is already in use through research partnerships in Nigeria, India, Pakistan and Rwanda. Dr Rosemary Townsend, an honorary consultant at the University of Edinburgh, stated: “The day of birth should be a joyful one, but it is also a time where mothers and babies can be vulnerable to devastating complications. The ANNE® Maternal system uses low-profile wearable sensors to allow birthing mothers comfort, mobility, and comprehensive state-of-the-art vital signs monitoring.”
Following the FDA clearance, Sibel will receive an additional 5 million dollar grant to work on AI-powered features. These tools are being designed to help clinicians in settings with low staff-to-patient ratios to detect potential risks earlier.