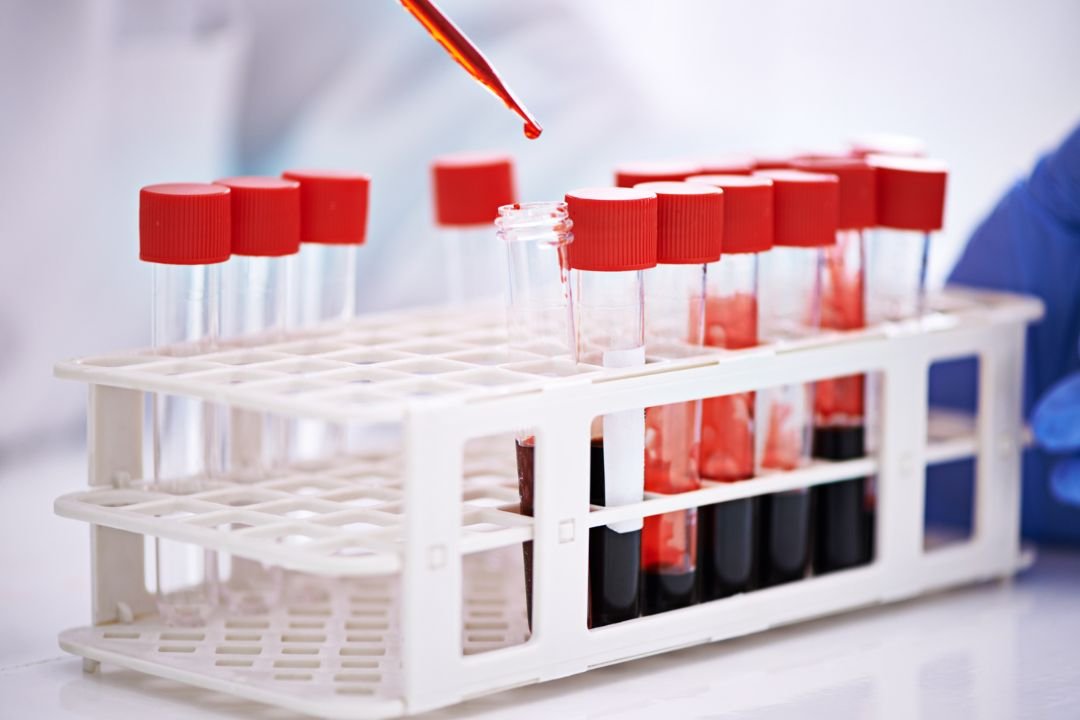
In a quiet corner of Nara Medical University, researchers are developing a breakthrough that could transform how the world responds to medical emergencies. A new form of artificial blood, no blood type matching required and capable of lasting two years in storage, is currently undergoing clinical trials in Japan.
The synthetic blood, created in the lab of Professor Hiromi Sakai, is being tested on volunteers who are receiving between 100 and 400 milliliters each. The effort aims to solve longstanding challenges in trauma care, surgery, and blood shortages, challenges made worse by the brief shelf life and blood type requirements of current donations.
Researchers extract hemoglobin from expired donated blood and encase it in a virus-resistant shell. This not only gives a second life to what would have been discarded but also removes the need for type compatibility, a major logistical barrier in emergency medicine.
The urgency of this work cannot be overstated. Blood transfusions are central to saving lives after accidents, during surgeries, and in childbirth. Yet, donation rates remain low worldwide, and the systems that rely on them constantly face shortages.
Complementing the research at Nara Medical University, Professor Teruyuki Komatsu of Chuo University is working on artificial oxygen carriers made using albumin, a common protein found in plasma. These carriers are being developed to help stabilize blood pressure and support treatment for strokes and hemorrhages.
This coordinated approach could dramatically improve emergency response, especially in places where donated blood isn’t readily available. The artificial blood’s potential for wide distribution and extended shelf life could make it a global solution by the end of the decade.
Japan could begin rolling out this innovation nationwide by 2030, pending the success of current trials. If successful, this may mark one of the most important shifts in medical care since the advent of antibiotics, not just in Japan, but worldwide.